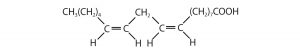
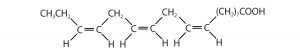
Saturated and Unsaturated Fatty Acids
Fatty acids are carboxylic acids that are structural components of fats and oils. More than 70 have been identified in nature. They usually contain an even number of carbon atoms (typically 12–20), are generally unbranched, and can be classified by the presence and number of carbon-to-carbon double bonds. Thus, saturated fatty acids contain no carbon-to-carbon double bonds, monounsaturated fatty acids contain one carbon-to-carbon double bond, and polyunsaturated fatty acids contain two or more carbon-to-carbon double bonds. The following table (“Some Common Fatty Acids Found in Natural Fats”) lists some common fatty acids and one important source for each.
Some Common Fatty Acids Found in Natural Fats
| Name | Abbreviated Chemical Formula | Condensed Structural Formula | Melting Point (°C) | Source |
| lauric acid | C11H23COOH | CH3(CH2)10COOH | 44 | palm kernel oil |
| myristic acid | C13H27COOH | CH3(CH2)12COOH | 58 | oil of numeg |
| palmitic acid | C15H31COOH | CH3(CH2)14COOH | 63 | palm oil |
| palmitoleic acid | C15H29COOH | CH3(CH2)5CH=CH(CH2)7COOH | 0.5 | macadamia oil |
| stearic acid | C17H35COOH | CH3(CH2)16COOH | 70 | cocoa butter |
| oleic acid | C17H33COOH | CH3(CH2)7CH=CH(CH2)7COOH | 16 | olive oil |
| linoleic acid | C17H31COOH | CH3(CH2)3(CH2CH=CH)2(CH2)7COOH | -5 | canola oil |
| α-linolenic acid | C17H29COOH | CH3(CH2CH=CH)3(CH2)7COOH | -11 | flaxseed |
| arachidonic acid | C19H31COOH | CH3(CH2)4(CH2CH=CH)4(CH2)2COOH | -50 | liver |
Cis and Trans Unsaturated Fatty Acids
In saturated fatty acids, there is free rotation about the carbon-carbon single bonds (C–C). In contrast, the carbon-carbon double bonds (C=C) in unsaturated fatty acids are rigid; rotation about doubly bonded carbon atoms is not possible without rupturing the bond. The figure below demonstrates the difference in rotation and rigidity between C-C bonds and C=C bonds.
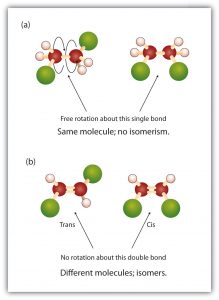
In the molecule in part (a) of the figure above, there is free rotation about the C–C bond. The two models shown represent exactly the same molecule; they are not isomers.
In the molecule in part (b) of the figure above, however, restricted rotation about the double bond means that the relative positions of substituent groups above or below the double bond become significant. This leads to a special kind of isomerism. The isomer in which the two green atoms lie on the same side of the molecule is called the cis isomer (Latin cis, meaning “on this side”). The isomer with the two green atoms on opposite sides of the molecule is the trans isomer (Latin trans, meaning “across”). These two compounds are cis-trans isomers (or geometric isomers), compounds that have different configurations (groups permanently in different places in space) because of the presence of a rigid structure in their molecule.
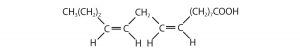
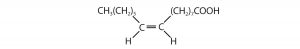
Consider the molecule in the figure below with the condensed structural formula CH3CH=CHCH3. There are actually two compounds with this formula; the double bond results in cis-trans isomerism. The cis isomer has both carbon atoms on the same side of the double bond. The trans isomer has both carbon atoms on opposite sides of the double bond. Their structural formulas are as follows:
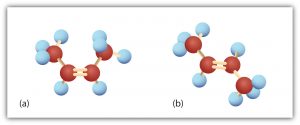
There are two possible configurations around the C=C bonds in unsaturated fatty acids due to the rigidity of C=C bond(s). The hydrocarbon chain around the double bonds in unsaturated fatty acids can be arranged in either the cis or trans isomeric form.
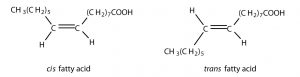
Naturally occurring fatty acids are generally in the cis configuration.
Structures and Properties Fatty Acids
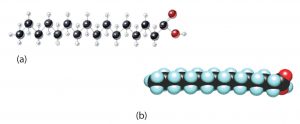
Although we often draw the carbon atoms in a straight line, they actually have more of a zigzag configuration (part (a) of the figure below). Viewed as a whole, however, the saturated fatty acid molecule is relatively straight (part (b) of the figure below). Such molecules pack closely together into a solid crystal lattice, maximizing the strength of attractive forces between the molecules and causing saturated fatty acids and the fats derived from them to have relatively high melting points. Most saturated fatty acids are solids at room temperature.
In contrast, each cis carbon-carbon double bond in an unsaturated fatty acid produces a pronounced bend in the molecule, so that these molecules do not stack neatly. As a result, the attractive forces between unsaturated fatty acids (and unsaturated fats) are weaker, causing these substances to have lower melting points. Most unsaturated fatty acids are liquids at room temperature.
Essential Fatty Acids
Two polyunsaturated fatty acids—linoleic and α-linolenic acids—are termed essential fatty acids because humans must obtain them from their diets. Both substances are required for normal growth and development, but the human body does not synthesize them. The body uses linoleic acid to synthesize many of the other unsaturated fatty acids, such as arachidonic acid. In addition, the essential fatty acids are necessary for the efficient transport and metabolism of cholesterol. The average daily diet should contain about 4–6 g of the essential fatty acids.
Concept Review Exercises
- Why do unsaturated fatty acids have lower melting points than saturated fatty acids?
-
Arrange these fatty acids (all contain 18 carbon atoms) in order of increasing melting point. Justify your arrangement.
-
Arrange these fatty acids (all contain 16 carbon atoms) in order of increasing melting point. Justify your arrangement.
- CH3(CH2)14COOH
Solutions
- Unsaturated fatty acids cannot pack as tightly together as saturated fatty acids due to the presence of the cis double bond that puts a “kink” or bend in the hydrocarbon chain.
-
3 < 1 < 2; an increase in the number of double bonds will lower the melting point because it is more difficult to closely pack the fatty acids together.
- 2 < 3 < 1; an increase in the number of double bonds will lower the melting point because it is more difficult to closely pack the fatty acids together.
Attributions
This page is based on “Chemistry 2e” by Paul Flowers, Klaus Theopold, Richard Langley, William R. Robinson, PhD, Openstax which is licensed under CC BY 4.0. Access for free at https://openstax.org/books/chemistry-2e/pages/1-introduction
This page is based on “The Basics of General, Organic, and Biological Chemistry” by David W Ball, John W Hill, Rhonda J Scott, Saylor which is licensed under CC BY-NC-SA 4.0. Access for free at http://saylordotorg.github.io/text_the-basics-of-general-organic-and-biological-chemistry/index.html