Peptides
Two or more amino acids can join together into chains called peptides. The -NH3+ group on one end of an amino acid molecule reacts with the -COO– group on another, releasing a molecule of water and forming a new bond:
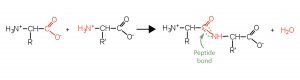 The linkage joining two amino acid units is called a peptide bond. Note that the product molecule still has a reactive -NH3+ group on the left and a reactive -COO– group on the right. These can react with additional amino acids to lengthen the peptide. The process can continue until thousands of units have joined, resulting in large proteins.
The linkage joining two amino acid units is called a peptide bond. Note that the product molecule still has a reactive -NH3+ group on the left and a reactive -COO– group on the right. These can react with additional amino acids to lengthen the peptide. The process can continue until thousands of units have joined, resulting in large proteins.
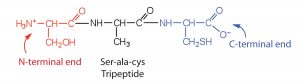
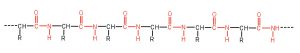 A chain consisting of only two amino acid units is called a dipeptide; a chain consisting of three is a tripeptide. For example, the following tripeptide consists of three amino acid units: serine, alanine, and cysteine. The amino acids are listed in order using their three-letter abbreviations: ser-ala-cys
A chain consisting of only two amino acid units is called a dipeptide; a chain consisting of three is a tripeptide. For example, the following tripeptide consists of three amino acid units: serine, alanine, and cysteine. The amino acids are listed in order using their three-letter abbreviations: ser-ala-cys
 The general term peptide refers to an amino acid chain of unspecified length. However, chains of about 50 amino acids or more are usually called proteins or polypeptides. In its physiologically active form, a protein may be composed of one or more polypeptide chains.
The general term peptide refers to an amino acid chain of unspecified length. However, chains of about 50 amino acids or more are usually called proteins or polypeptides. In its physiologically active form, a protein may be composed of one or more polypeptide chains.
For peptides and proteins to function correctly, it is not enough that they incorporate certain amounts of specific amino acids. The order, or sequence, in which the amino acids are connected is also of critical importance. Bradykinin is a nine-amino acid peptide produced in the blood that has the following amino acid sequence:
arg-pro-pro-gly-phe-ser-pro-phe-arg
This peptide lowers blood pressure, stimulates smooth muscle tissue, increases capillary permeability, and causes pain. When the order of amino acids in bradykinin is reversed,
arg-phe-pro-ser-phe-gly-pro-pro-arg
the peptide resulting from this synthesis shows none of the activity of bradykinin.
Just as millions of different words are spelled with our 26-letter English alphabet, millions of different proteins are made with the 20 common amino acids. However, just as the English alphabet can be used to write gibberish, amino acids can be put together in the wrong sequence to produce nonfunctional proteins. Although the correct sequence is ordinarily of utmost importance, it is not always absolutely required. Just as you can sometimes make sense of incorrectly spelled English words, a protein with a small percentage of “incorrect” amino acids may continue to function. However, it rarely functions as well as a protein having the correct sequence. There are also instances in which seemingly minor errors of sequence have disastrous effects. For example, in some people, every molecule of hemoglobin (a protein in the blood that transports oxygen) has a single incorrect amino acid unit out of about 300 (a single valine replaces a glutamic acid). That “minor” error is responsible for sickle cell anemia, an inherited condition that usually is fatal.
Concept Review Exercise
Describe the difference between an amino acid and a peptide.
Solution
A peptide is composed of two or more amino acids. Amino acids are the building blocks of peptides
Attributions
This page is based on “Chemistry 2e” by Paul Flowers, Klaus Theopold, Richard Langley, William R. Robinson, PhD, Openstax which is licensed under CC BY 4.0. Access for free at https://openstax.org/books/chemistry-2e/pages/1-introduction
This page is based on “The Basics of General, Organic, and Biological Chemistry” by David W Ball, John W Hill, Rhonda J Scott, Saylor which is licensed under CC BY-NC-SA 4.0. Access for free at http://saylordotorg.github.io/text_the-basics-of-general-organic-and-biological-chemistry/index.html

