Element Names and Symbols
Just as a language has an alphabet from which words are built, chemistry has an alphabet from which matter is described. However, the chemical alphabet is larger than the one we use for spelling. The chemical alphabet consists of the 118 elements on the periodic table that have been discovered so far. An element is a substance that cannot be broken down into simpler chemical substances. Their role is central to chemistry, for they combine to form the millions and millions of known compounds.
Names and Symbols
Each element has a name. Some of these names date from antiquity, while others are quite new. Today, the names for newly discovered elements are proposed by their discoverers but must be approved by the International Union of Pure and Applied Chemistry (IUPAC), an international organization that makes recommendations concerning all kinds of chemical terminology.
The names of the elements can be cumbersome to write in full, especially when combined to form the names of compounds. Therefore, each element name is abbreviated as a one- or two-letter chemical symbol. By convention, the first letter of a chemical symbol is a capital letter, while the second letter (if there is one) is a lowercase letter. The first letter of the symbol is usually the first letter of the element’s name, while the second letter is some other letter from the name. Some elements have symbols that derive from earlier, mostly Latin names, so the symbols may not contain any letters from the English name.
For example, the symbol for mercury is Hg. It is derived from mercury’s Latin name, hydrargyrum. We use the same symbol to indicate one atom of mercury (microscopic domain) or to label a container of many atoms of the element mercury (macroscopic domain).
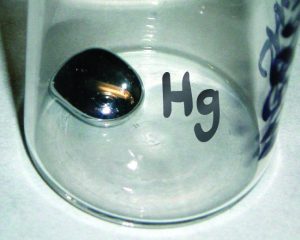
The following table lists the names and symbols of some common elements.
| Element | Symbol | Element | Symbol |
|---|---|---|---|
| aluminum | Al | iron | Fe (from ferrum) |
| bromine | Br | lead | Pb (from plumbum) |
| calcium | Ca | magnesium | Mg |
| carbon | C | mercury | Hg (from hydrargyrum) |
| chlorine | Cl | nitrogen | N |
| chromium | Cr | oxygen | O |
| cobalt | Co | potassium | K (from kalium) |
| copper | Cu (from cuprum) | silicon | Si |
| fluorine | F | silver | Ag (from argentum) |
| gold | Au (from aurum) | sodium | Na (from natrium) |
| helium | He | sulfur | S |
| hydrogen | H | tin | Sn (from stannum) |
| iodine | I | zinc | Zn |
Element names in languages other than English are often close to their Latin names. For example, gold is oro in Spanish and or in French (close to the Latin aurum), tin is estaño in Spanish (compare to stannum), lead is plomo in Spanish and plomb in French (compare to plumbum), silver is argent in French (compare to argentum), and iron is fer in French and hierro in Spanish (compare to ferrum). The closeness is even more apparent in pronunciation than in spelling.
Properties of Elements
Most elements in their pure form exist as individual atoms. For example, a macroscopic chunk of iron metal is composed, microscopically, of individual atoms. Some elements, however, exist as groups of atoms called molecules, as discussed in Chapter 1 “Atoms and Molecules.” Several important elements exist as two-atom combinations and are called diatomic molecules. In representing a diatomic molecule, we use the symbol of the element and include the subscript 2 to indicate that two atoms of that element are joined together. The elements that exist as diatomic molecules are hydrogen (H2), oxygen (O2), nitrogen (N2), fluorine (F2), chlorine (Cl2), bromine (Br2), and iodine (I2).
While many elements differ dramatically in their chemical and physical properties, some elements have similar properties. For example, many elements conduct heat and electricity well, whereas others are poor conductors. These properties can be used to sort the elements into three classes: metals (elements that conduct well), nonmetals (elements that conduct poorly), and metalloids (elements that have intermediate conductivities).
The periodic table is a table of elements that places elements with similar properties close together. You will learn more about the periodic table as you continue your study of chemistry.
Attributions
This page is based on “Chemistry 2e” by Paul Flowers, Klaus Theopold, Richard Langley, William R. Robinson, PhD, Openstax which is licensed under CC BY 4.0. Access for free at https://openstax.org/books/chemistry-2e/pages/1-introduction
This page is based on “Chemistry of Cooking” by Sorangel Rodriguez-Velazquez which is licensed under CC BY-NC-SA 4.0. Access for free at http://chemofcooking.openbooks.wpengine.com/
This page is based on “The Basics of General, Organic, and Biological Chemistry” by David W Ball, John W Hill, Rhonda J Scott, Saylor which is licensed under CC BY-NC-SA 4.0. Access for free at http://saylordotorg.github.io/text_the-basics-of-general-organic-and-biological-chemistry/index.html

